The global Transcutaneous Electrical Nerve Stimulation (TENS) stimulator market has undergone a significant transformation. As chronic pain becomes a widespread global health issue—affecting nearly 20% of the world's population—the demand for non-pharmacological, drug-free pain management solutions has skyrocketed. Today, TENS stimulators are no longer limited to clinical settings; they have become essential tools in sports medicine, home rehabilitation, and geriatric care.
Globally, North America and Europe remain the largest markets due to advanced healthcare infrastructures and a high awareness of physiotherapy. However, the Asia-Pacific region, led by China's manufacturing prowess, is the fastest-growing hub for both production and consumption. The integration of AI-driven protocols and wireless connectivity has shifted the industry from simple pulse generators to sophisticated, data-logging medical devices.
Expanding across 120+ countries, providing critical relief for orthopedic and neurological recovery.
Modern stimulators now utilize machine learning to adjust pulse frequency based on real-time muscle feedback, optimizing recovery times.
Transitioning from wired units to Bluetooth-enabled wearable stimulators controlled via smartphone applications for user convenience.
Combining TENS with EMS (Electrical Muscle Stimulation) and heat therapy in a single portable device for comprehensive rehabilitation.
As a leading China TENS Stimulator manufacturer, Xinkang Biomedical is at the forefront of these trends. We emphasize the transition toward biofeedback-integrated systems. Our R&D team focuses on "Smart Rehabilitation," ensuring that our devices can track patient progress and provide clinical-grade data to healthcare providers remotely.
Years Experience
OEM Customers
China Hospital Coverage
Factory Area (sqm)
Partnering with a China TENS Stimulator supplier like Xinkang Biomedical offers unparalleled advantages in the global supply chain. Located in the Yangtze River Delta, we benefit from a complete industrial ecosystem, allowing us to source high-quality components at competitive costs.
Our facility houses 30+ standard OEM production lines, ensuring we can fulfill bulk procurement orders with short lead times. Whether you are a medical equipment distributor in London or a rehabilitation chain in New York, our localized support and flexible manufacturing (OEM/ODM) empower your brand to lead in your local market.

Focused on the research, development, and manufacturing of rehabilitation medical equipment for 15 years, integrating R&D, production, and sales to create a full-cycle closed loop. We are a domestic leader in physical factor treatment products such as electrotherapy, light therapy, magnetic therapy, pressure therapy, and spinal and sports therapy products.
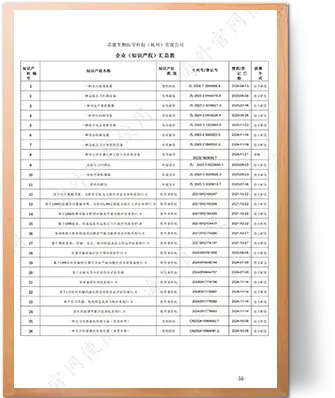
1 Invention Patent | 3 Design Patents | 7 Utility Model Patents | 13 Software Copyrights | 10 Registered Trademarks
Our R&D personnel are from top domestic and foreign universities. Software and hardware are independently developed, ensuring safe and localized data storage.
Passed national medical supervision and management certification. We use international standards as our benchmark for every process.
Factory covers over 10,000 square meters with 200+ employees and 30+ standard OEM production lines.




Our OEM R&D team consists of engineers with over 10 years of experience in rehabilitation medical equipment. We provide product training, sales courses, assembly guidance, and 24/7 technical support to ensure our global partners succeed.
Covering more than 75% of tertiary medical units and most rehabilitation clinical departments in China. We collaborate with top universities and research centers.

Actively building a systematic global development plan to bring high-quality Chinese rehabilitation technology to the international stage.




