 OEM Advanced magnetic stimulation therapy equipment for rehabilitation Smart Poor-Handling Robot Factories
OEM Advanced magnetic stimulation therapy equipment for rehabilitation Smart Poor-Handling Robot Factories
 OEM Shock Wave Therapy Instrument (Pneumatic Ballistic) Manufacturer, Supplier
OEM Shock Wave Therapy Instrument (Pneumatic Ballistic) Manufacturer, Supplier
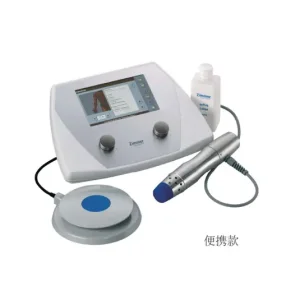 China Shock Wave Therapy Instrument (Electromagnetic Ballistic Type) Manufacturers
China Shock Wave Therapy Instrument (Electromagnetic Ballistic Type) Manufacturers
 OEM Smart robotic fascia release device for muscle recovery 3D Insole Printer
OEM Smart robotic fascia release device for muscle recovery 3D Insole Printer
 China Multi-position Medical Diagnosis and Treatment Bed (9 Sections) Manufacturers
China Multi-position Medical Diagnosis and Treatment Bed (9 Sections) Manufacturers
 High-Quality Ankle Continuous Passive Training Instrument Manufacturer
High-Quality Ankle Continuous Passive Training Instrument Manufacturer
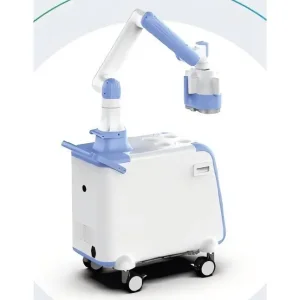 High-Quality Automated moxibustion therapy robot for traditional Chinese rehabilitation
High-Quality Automated moxibustion therapy robot for traditional Chinese rehabilitation
 OEM Transcranial Electric Stimulator (Tes) Wireless Transcranial Electrical Stimulator
OEM Transcranial Electric Stimulator (Tes) Wireless Transcranial Electrical Stimulator
The global market for Transcutaneous Electrical Nerve Stimulation (TENS) devices has seen exponential growth over the last decade. As chronic back pain becomes a leading cause of disability worldwide, affecting over 600 million people, the demand for non-invasive, drug-free pain management solutions has skyrocketed. Modern TENS devices are no longer bulky clinical machines; they have evolved into wearable, smart-integrated technology that allows patients to manage pain during daily activities.
Industry reports suggest that the rehabilitation equipment market is shifting toward home-based care and telehealth integration. Suppliers and manufacturers in the TENS space are now focusing on precision—targeting specific nerve paths associated with lumbar and cervical pain with high clinical accuracy. As a leading TENS device manufacturer, Xinkang Biomedical is at the forefront of this industrial revolution, merging medical expertise with AI-driven hardware.
The CAGR for pain management devices is projected to stay above 6.5% through 2030, driven by an aging population and rising sports injuries.
Compliance with FDA, CE, and ISO 13485 is now the baseline for global procurement, ensuring patient safety and device reliability.
Large-scale medical distributors are seeking long-term OEM partners to fulfill the massive inventory needs of private hospitals and rehab centers.
Focused on the research, development, and manufacturing of rehabilitation medical equipment for 15 years, integrating R&D, production, and sales to create a full-cycle closed loop. We are a domestic leader in physical factor treatment products such as electrotherapy, light therapy, magnetic therapy, and spinal therapy.

The modern procurement trend is moving away from wired units. Global buyers are demanding wireless, app-controlled TENS devices that can be worn under business attire. This localization of "therapy on the go" is a key selling point for distributors in North America and Europe.
Advanced TENS devices now incorporate AI to analyze skin impedance and adjust pulse widths automatically. This "Smart Stimulation" prevents muscle habituation and ensures long-term efficacy for chronic back pain sufferers.
Hospitals are looking for devices that offer both TENS (Pain relief) and EMS (Muscle stimulation) in one unit. Our multi-channel systems allow for simultaneous treatment of the lower back and shoulder areas, optimizing clinical time.
Where our TENS technology makes a difference every day.
Used by physiotherapists to manage post-surgical back pain and facilitate faster patient discharge through non-opioid pain control.
Empowering the elderly and those with sedentary lifestyles to manage sciatica and lumbar strain from the comfort of their sofa.
Professional athletes utilize our high-frequency stimulation for rapid muscle recovery and pain relief after intensive training sessions.





Our product technology R&D personnel are all from top domestic and foreign universities. The software and hardware are independently developed and designed; diversified data storage and simple, fast, convenient and safe localization.
Our products have covered more than 75% of tertiary medical units and most rehabilitation clinical departments. We cooperate with colleges, research centers, and medical centers.
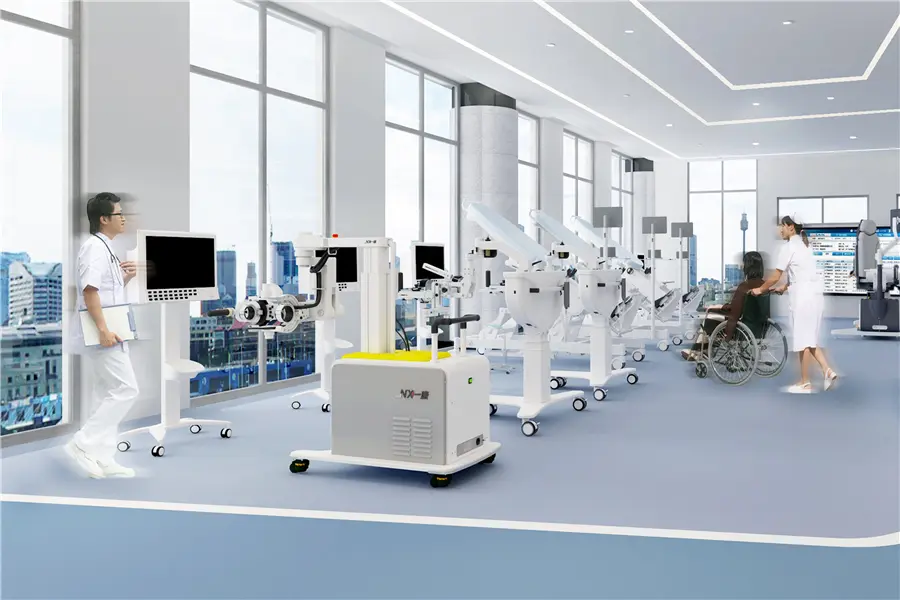
We are expanding our high-quality medical device footprints globally, offering OEM production, assembly guidance, and technical after-sales maintenance to partners worldwide.





 China Professional ultrasonic therapy device for pain relief Foot Scanner Manufacturer
China Professional ultrasonic therapy device for pain relief Foot Scanner Manufacturer
 OEM 3D Gait Analysis System Suppliers, Factory
OEM 3D Gait Analysis System Suppliers, Factory
 OEM Upper and Lower Limb Active and Passive Training System Manufacturer
OEM Upper and Lower Limb Active and Passive Training System Manufacturer
 China Portable Version of 3D Spine Posture Measurement Supplier
China Portable Version of 3D Spine Posture Measurement Supplier
 China Trackless Muscle Strength Training and Evaluation System Supplier
China Trackless Muscle Strength Training and Evaluation System Supplier
 OEM Hand-Held High Frequency Therapeutic Apparatus Suppliers
OEM Hand-Held High Frequency Therapeutic Apparatus Suppliers
 OEM Dynamic/Static Balance Function Assessment and Training System Suppliers
OEM Dynamic/Static Balance Function Assessment and Training System Suppliers
 China Wearable walking assist robot for regaining independent gait training Manufacturer
China Wearable walking assist robot for regaining independent gait training Manufacturer
Choosing a TENS Device for Back Pain Manufacturer requires more than just looking at a price tag. It requires a partner with deep scientific expertise. At Xinkang, our engineers focus on the Gate Control Theory of Pain and Endorphin Release protocols. By optimizing the frequency (Hz) and pulse width (µs) of our devices, we provide therapeutic grades of relief that surpass consumer-level electronics found on mass marketplaces.
Our commitment to E-E-A-T (Experience, Expertise, Authoritativeness, and Trustworthiness) is reflected in our 30-year legacy and our continuous collaboration with top medical universities. Whether you are a local clinic in the UK, a distributor in the USA, or a hospital in the Middle East, our localized support and Chinese manufacturing prowess provide you with the competitive edge needed in today's medical device market.