The Lumbar Traction Device (Saunders style) has become the gold standard in non-surgical spinal decompression worldwide. As chronic lower back pain affects nearly 10% of the global population, the demand for high-precision, force-accurate traction systems has skyrocketed. In the current global landscape, medical facilities are shifting from expensive, bulky traction beds to more portable, patient-operated Saunders-type units that provide clinical-grade results.
Industry reports indicate a significant CAGR of 6.8% in the spinal rehabilitation sector. This growth is driven by a preference for Active Spinal Decompression over traditional methods. Factories in China, particularly those focused on medical biomedical technology, are now leading the charge in refining these pneumatic and mechanical systems to ensure safety and therapeutic efficacy.

Modern Saunders devices utilize high-grade pneumatic cylinders that ensure smooth force delivery, preventing muscle guarding and maximizing spinal opening.
The latest trend involves Bluetooth-connected sensors that track traction force and duration, allowing clinicians to monitor patient progress remotely.
Ergonomic contouring ensures that pelvic and thoracic belts distribute force evenly, reducing localized pressure and enhancing patient comfort.
Procurement teams from North America, Europe, and the Middle East are increasingly looking for China Lumbar Traction Device Saunders Factories due to the unparalleled balance of cost-effectiveness and high-quality standards. The procurement needs vary by region:
Xinkang Biomedical Technology addresses these localized needs by offering customizable OEM/ODM solutions that comply with regional medical standards, including ISO and CE certifications.
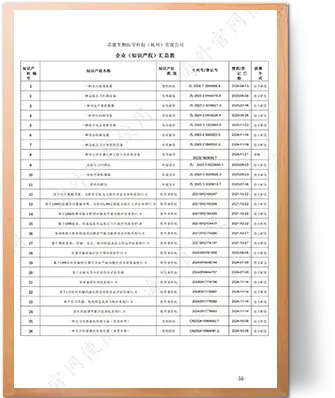
Focused on the research, development, and manufacturing of rehabilitation medical equipment for 15 years as a specialized brand, within a 30-year industrial history.
Located in the heart of China's advanced manufacturing hub, Xinkang Biomedical integrates a complete supply chain. From software development to precision hardware casting, every component of our Lumbar Traction Device is quality-controlled within our 10,000+ square meter facility.
Our commitment to innovation is backed by:
- 1 Invention Patent
- 3 Design Patents
- 7 Utility Model Patents
- 13 Software Copyrights
Our engineers bring over 10 years of experience in rehabilitation technology, focusing on technology integration and market insights to lead the industry.
We utilize international peer quality standards as our baseline, ensuring every device undergoes rigorous testing before leaving the assembly line.









Xinkang Biomedical Technology (Hangzhou) Co., Ltd. is committed to improving people's quality of life through innovation in pain rehabilitation, orthopedic rehabilitation, and neurological rehabilitation. We are a domestic leader in physical factor treatment products such as electrotherapy, light therapy, magnetic therapy, and spinal therapy.