Advanced Low Frequency & Physical Therapy Solutions for Global Clinical Excellence

OEM Shock Wave Therapy Instrument (Pneumatic Ballistic) Manufacturer, Supplier

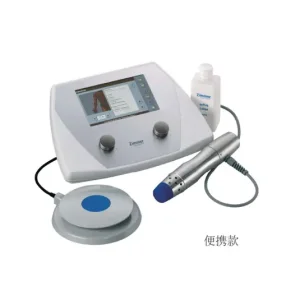
China Shock Wave Therapy Instrument (Electromagnetic Ballistic Type) Manufacturers, Factories

High-Quality Ankle Continuous Passive Training Instrument Manufacturer, Factory
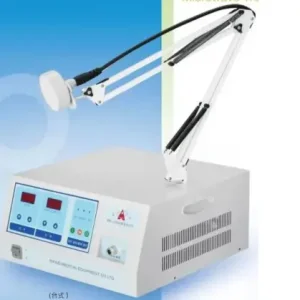

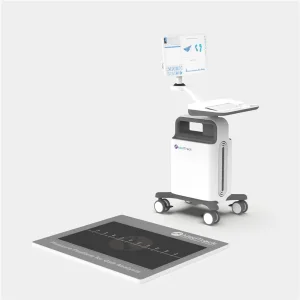
OEM Plantar Pressure Gait Evaluation System Supplier, Factory

The global low frequency therapy device market is witnessing an unprecedented CAGR of 6.8%, driven by an aging population and a shift toward non-invasive pain management. From Transcutaneous Electrical Nerve Stimulation (TENS) to Electrical Muscle Stimulation (EMS), these technologies are no longer confined to hospitals; they are becoming essential components of home-based rehabilitation and athletic recovery programs worldwide.
The industry is moving toward Miniaturization, Intelligence, and Connectivity. Wearable low frequency devices integrated with AI biofeedback are the current frontier. Modern suppliers are focusing on "Smart Therapy"—devices that can adjust pulse width and frequency automatically based on the user's neurological response, ensuring maximum therapeutic efficacy with minimal discomfort.
Global buyers, particularly from North America and Europe, are demanding more than just low prices. The focus has shifted to Medical Grade Compliance (FDA/CE), stable supply chains, and robust OEM/ODM capabilities. Procurement officers are looking for partners who provide full-cycle support, from industrial design to technical after-sales service.
Years Experience
Market Coverage
OEM Partners
Focused on the research, development, and manufacturing of rehabilitation medical equipment for 15 years, integrating R&D, production, and sales to create a full-cycle closed loop. We are the most professional pain management expert, deeply engaged in the field of rehabilitation medicine.

At Xinkang Medical, our R&D technology leads the domestic industry. With personnel from top global universities, we develop both software and hardware independently. Our 10,000㎡ factory in the Yangtze River Delta ensures a complete supply chain, supporting over 30 standard OEM production lines.
The company has:
1 invention patent | 3 design patents | 7 utility model patents | 13 software copyrights | 10 registered trademarks



Tailored Solutions for Different Professional Environments
Used in neurological rehabilitation departments for stroke recovery, muscle atrophy prevention, and chronic pain management (lower back pain, arthritis).
Elite sports centers utilize our EMS systems for rapid lactic acid clearance and localized muscle strengthening for professional athletes.
Compact, handheld low frequency devices designed for elderly care, allowing patients to continue therapy sessions in a comfortable domestic setting.
Choosing a China Low Frequency Therapy Device Supplier like Xinkang Biomedical offers strategic advantages in the global marketplace. The Chinese medical device industry has matured into a global powerhouse, combining cost-efficiency with high-tier technological integration. Our factory, located in Hangzhou, benefits from the "Silicon Valley of the East" ecosystem, providing us access to the latest IoT and AI hardware components.
Our location in the Yangtze River Delta means every component—from the PCB boards to the medical-grade silicone electrodes—is sourced within a 100km radius. This drastically reduces lead times and allows us to offer unbeatable price-to-performance ratios compared to European or American manufacturers.
We understand that different markets have different ergonomic and linguistic requirements. Our R&D team offers personalized customized services including equipment housing design, UI software localization, and branded marketing materials. With over 30 production lines, we can handle small-batch prototypes and mass-market rollouts simultaneously.
Adhering to international standards is our baseline. We implement a multi-stage QC process: raw material inspection, SMT (Surface Mount Technology) testing, assembly line verification, and a 48-hour aging test for every electronic therapeutic device. This ensures that the Low Frequency Therapy Device delivered to your warehouse is ready for immediate clinical use.







Empowering Rehabilitation with Intelligent Data and Robotics

China Portable Version of 3D Spine Posture Measurement Supplier, Factory



High-Quality Upper Limb Intelligent Feedback Training System Manufacturer, Supplier

OEM Upper and Lower Limb Active and Passive Training System Manufacturer, Factories

High-Quality Multi-joint Isokinetic Training and Testing System Manufacturers, Factory

China Trackless Muscle Strength Training and Evaluation System Supplier, Suppliers
As we look toward 2030, the Low Frequency Therapy Device industry is poised for a digital revolution. "Cloud-based Physical Therapy" will allow doctors to prescribe specific pulse patterns remotely, which the patient's device can then download and execute. At Xinkang Biomedical, we are already integrating Bluetooth connectivity and cloud diagnostics into our newest product lines.
Our commitment to E-E-A-T principles ensures that every piece of equipment is backed by clinical research and 30 years of manufacturing wisdom. Whether you are a distributor looking for a reliable factory or a medical center seeking high-precision instruments, we provide the authoritative solutions needed to improve patient outcomes and quality of life.