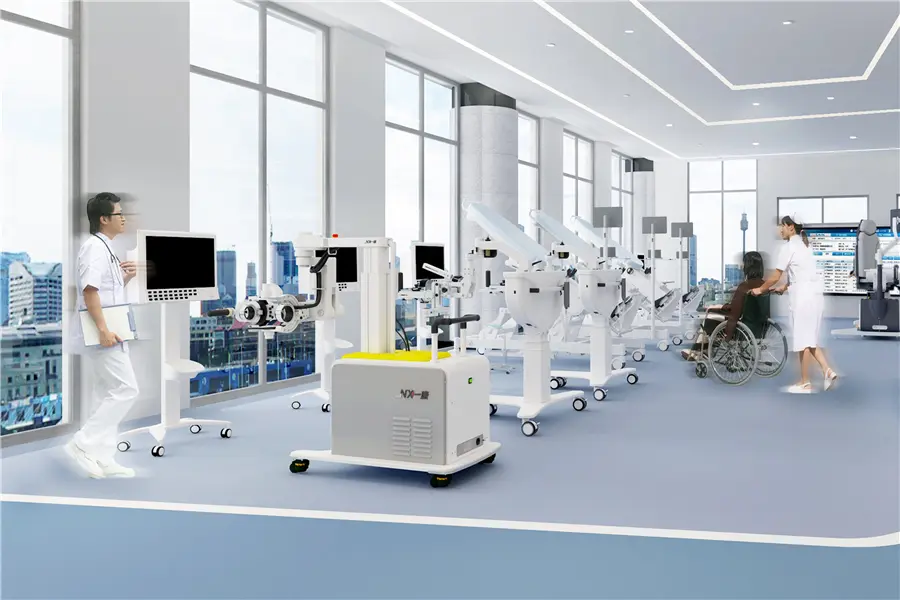
Professional Grade Rehabilitation & Stimulation Equipment
 OEM Advanced Magnetic Stimulation Therapy Equipment
OEM Advanced Magnetic Stimulation Therapy Equipment
 Compound Wave Dynamic Interference Electrotherapy
Compound Wave Dynamic Interference Electrotherapy
 Transcranial Electric Stimulator (TES) Wireless
Transcranial Electric Stimulator (TES) Wireless
 OEM 3D Gait Analysis System Suppliers
OEM 3D Gait Analysis System Suppliers
 Upper and Lower Limb Active/Passive Training
Upper and Lower Limb Active/Passive Training
 China Wearable Walking Assist Robot
China Wearable Walking Assist Robot
 Portable 3D Spine Posture Measurement
Portable 3D Spine Posture Measurement
 Diabetic Foot Dynamics Monitoring System
Diabetic Foot Dynamics Monitoring System
The global market for Electrical Neuromuscular Stimulators is experiencing an unprecedented surge, driven by an aging global population, the rising prevalence of chronic neurological disorders, and a paradigm shift toward non-invasive rehabilitation therapies. As a leading China Electrical Neuromuscular Stimulator supplier, we recognize that NMES technology has evolved from simple muscle toning devices to sophisticated medical-grade systems integrated with AI and biofeedback mechanisms.
CAGR 6.5%+
Steady growth in medical electronics
$1.2B Market
Projected size for neuro-rehab by 2028
Hospitals & Sports
Leading procurement segments
Today, the industrial status of neuromuscular stimulators is characterized by high-precision manufacturing. Tier-1 medical institutions in North America, Europe, and Asia are increasingly moving away from purely pharmacological interventions for muscle atrophy and motor dysfunction, favoring the functional restoration provided by NMES. This shift has catalyzed a massive demand for reliable NMES suppliers who can provide clinical-grade efficacy combined with user-friendly interfaces.
Modern stimulators are no longer static. They now incorporate AI algorithms that analyze muscle fatigue and recruitment patterns in real-time, adjusting current intensity to optimize therapeutic outcomes while ensuring patient comfort.
The industry is moving toward "rehab-on-the-go." Wireless electrodes and lightweight, wearable control units allow patients to undergo therapy while performing functional tasks, significantly accelerating the recovery process.
Telehealth integration allows clinicians to monitor patient progress remotely. Our latest stimulators export treatment data to secure cloud platforms, enabling data-driven adjustments to rehabilitation protocols.
International procurement officers and medical distributors are no longer looking for just a "product"; they are seeking integrated solutions. As a premier China Electrical Neuromuscular Stimulator manufacturer, we have identified key procurement priorities across different regions:
In hospital settings, the focus is on multi-channel versatility. Devices must be able to treat hemiplegia, spinal cord injuries, and post-operative muscle disuse atrophy simultaneously. Our systems offer multi-user modes and clinical presets that align with international medical standards.
Elite sports clinics require devices capable of high-intensity stimulation for muscle recovery and explosive strength training. The reliability of the pulse wave and the quality of the electrode interface are critical for these high-demand environments.
With the rise of the "Silver Economy," there is a massive localized demand for simplified, safe stimulators for home use. Localization involves translating interfaces into multiple languages and ensuring the hardware meets local safety certifications (CE, FDA, ISO).
Why partner with a China Electrical Neuromuscular Stimulator supplier? The answer lies in the unique synergy of the Chinese industrial ecosystem:

Focused on the research, development, and manufacturing of rehabilitation medical equipment for 15 years, integrating R&D, production, and sales to create a full-cycle closed loop.
Xinkang Biomedical Technology (Hangzhou) Co., Ltd. is a company committed to improving quality of life through innovation in pain rehabilitation, orthopedic rehabilitation, neurological rehabilitation, and mobile medical care. We are a domestic leader in physical factor treatment products including electrotherapy, light therapy, magnetic therapy, and spinal therapy.



Our products cover more than 75% of tertiary medical units and most rehabilitation clinical departments through systematic development and cooperation with research centers.
Expanding globally with high-quality assurance and international standards, serving over 5000 OEM customers worldwide.

Personnel from top domestic and foreign universities. Software and hardware are independently developed for simple, fast, and safe localization.
Factory over 10,000 sqm, 200+ employees, 30+ standard OEM production lines located in the core supply chain area.
Providing product training, sales courses, and full assembly guidance to ensure partner success.
 OEM 3D Gait Analysis System Factory
OEM 3D Gait Analysis System Factory
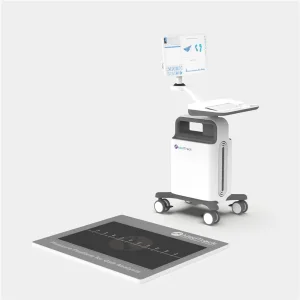 Plantar Pressure Gait Evaluation System
Plantar Pressure Gait Evaluation System
 Multi-joint Isokinetic Training System
Multi-joint Isokinetic Training System
 Dynamic/Static Balance Assessment System
Dynamic/Static Balance Assessment System
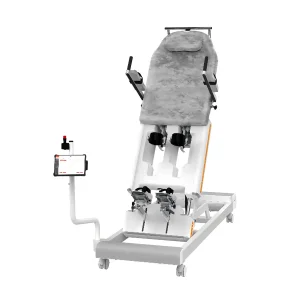 Intelligent Feedback Training for Lower Limbs
Intelligent Feedback Training for Lower Limbs
 Trackless Muscle Strength Training System
Trackless Muscle Strength Training System
 Active and Passive Training Manufacturers
Active and Passive Training Manufacturers
 Mobile Interference Electrotherapy Factory
Mobile Interference Electrotherapy Factory